A) soluble
B) polar
C) flammable
D) colored
F) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
The boiling point for normal alkanes will increase as the number of carbons increases.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Inorganic compounds are never found in living systems.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following formulas does not represent an organic compound?
A) CH4
B) CH3CH2NH2
C) CH3CH3CH3
D) All are organic compounds
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is not correct? Functional groups are
A) to organic chemistry as polyatomic ions are to inorganic chemistry.
B) the parts of molecules used to differentiate classifications of organic molecules.
C) are chemically bound to each other in order to develop larger molecules.
D) there is more than one correct response.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following hydrocarbon reacts with an excess of oxygen gas,the balanced equation would contain how many moles of water? 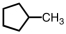
A) 5
B) 6
C) 10
D) 12
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following formulas represents a saturated hydrocarbon?
A) C2H6
B) C2H4
C) C3H6
D) C5H10
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structural formulas represents an normal (unbranched) alkane?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Since all alkanes are saturated hydrocarbons,all of the carbon atoms are aligned in one continuous chain.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which applies to the manner in which carbon bonds in alkanes?
A) The bonds are formed with carbon using an sp3 configuration.
B) The bonds between carbon and other atoms are ionic.
C) Carbon to carbon bonds are expected to be double bonds.
D) The bonds formed by carbon all use p orbitals.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Isomers always have the same molecular formula.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following two structures would be classified as what? 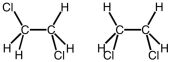
A) geometric isomers
B) structural isomers
C) stereoisomers
D) conformations
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following compound would be named ____-1,2-dimethylcyclopropane. 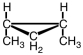
A) cis
B) trans
C) either cis or trans
D) neither cis nor trans
F) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Most organic compounds contain ionic bonds.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Alkanes react readily with _____ .
A) sodium hydroxide
B) hydrochloric acid
C) sulfuric acid
D) none of the choices
F) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Isomers have identical physical and chemical properties.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many hydrogen atoms are needed to complete the following structure? C=C-N-C
A) 6
B) 7
C) 9
D) 10
F) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Isomers differ from each other in the number of neutrons present in each.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The name for the following structure will end with ____. 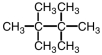
A) ethane
B) butane
C) hexane
D) octane
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Three physical properties shown below beside different alkane series.Which of the series is(are) listed in the order of increase (lowest to highest) for the corresponding property?
A) Density: C4H10 < C5H12 < C8H18
B) Melting Point: C8H18 < C5H12 < C4H10
C) Boiling Point: C5H12 < C4H10 < C8H18
D) All are correctly ranked.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 87
Related Exams