A) the ions,which are not present in pure water,balance the accumulation of charge in each of the half-cells.
B) the chloride ions increase the pH of the solution.
C) the sodium ions oxidize the iron atoms.
D) the chloride ions form a precipitate with iron.
E) the chloride ions oxidize the iron atoms.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given:
Li+(aq) + e-  Li(s) ; E° = -3.04 V
Mg2+(aq) + 2e-
Li(s) ; E° = -3.04 V
Mg2+(aq) + 2e-  Mg(s) ; E° = -2.38 V
Fe2+(aq) + 2e-
Mg(s) ; E° = -2.38 V
Fe2+(aq) + 2e-  Fe(s) ; E° = -0.41 V
Ag+(aq) + e-
Fe(s) ; E° = -0.41 V
Ag+(aq) + e-  Ag(s) ; E° = 0.80 V
Br2(l) + 2e-
Ag(s) ; E° = 0.80 V
Br2(l) + 2e-  2Br-(aq) ; E° = 1.07 V
Which of the following species is the best oxidizing agent?
2Br-(aq) ; E° = 1.07 V
Which of the following species is the best oxidizing agent?
A) Mg2+
B) Br-
C) Fe2+
D) Li
E) Ag+
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell is made by placing an iron electrode in a compartment in which the Fe2+ concentration is 3.0 × 10-5 M and by placing a Pt electrode in the other compartment,in which the H+ concentration is 3.10 M and 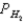 = 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is E for the cell at 25oC?
= 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is E for the cell at 25oC?
A) 0.10 V
B) 0.57 V
C) 0.27 V
D) 0.25 V
E) 0.56 V
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The cell potential of an electrochemical cell with the cell reaction 2Al(s) + 3Zn2+(aq) → 3Zn(s) + 2Al3+(aq) Is 1.607 V.What is the maximum electrical work obtainable from this cell when 2.0 mol of Al is consumed?
A) ![]() J
J
B) ![]() J
J
C) ![]() J
J
D) ![]() J
J
E) ![]() J
J
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A fuel cell designed to react grain alcohol with oxygen has the following net reaction: C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l) The maximum work that 1 mol of alcohol can yield by this process is 1320 kJ.What is the theoretical maximum voltage that this cell can achieve?
A) 1.14 V
B) 2.28 V
C) 2.01 V
D) 0.760 V
E) 13.7 V
G) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Balance the following half-reaction occurring in acidic solution.
NO3-(aq) → 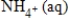
A) NO3-(aq) + 10H+(aq) + 8e− → ![]() + 3H2O(l)
+ 3H2O(l)
B) NO3-(aq) + 3H2O(l) + 10e− → ![]() + 10H+(aq)
+ 10H+(aq)
C) NO3-(aq) + 10H+(aq) → ![]() + 3H2O(l) + 10e−
+ 3H2O(l) + 10e−
D) NO3-(aq) + 8e− → ![]() + 8H+(aq) + 3H2O(l)
+ 8H+(aq) + 3H2O(l)
E) NO3-(aq) + 10H+(aq) → ![]() + 3H2O(l)
+ 3H2O(l)
G) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Given:
Zn2+(aq) + 2e-  Zn(s) ; E° = -0.76 V
Cu2+(aq) + 2e-
Zn(s) ; E° = -0.76 V
Cu2+(aq) + 2e-  Cu(s) ; E° = 0.34 V
Cr2O72-(aq) + 14H+(aq) + 6e-
Cu(s) ; E° = 0.34 V
Cr2O72-(aq) + 14H+(aq) + 6e-  2Cr3+(aq) + 7H2O(l) ; E° = 1.33 V
Which of the following species is the strongest oxidizing agent?
2Cr3+(aq) + 7H2O(l) ; E° = 1.33 V
Which of the following species is the strongest oxidizing agent?
A) Zn
B) Cr3+
C) Cr2O72-
D) Zn2+
E) Cu
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An alkaline dry cell is similar to Leclanché cell,but it has _____ in place of ammonium chloride.
A) potassium hydroxide
B) sodium hydroxide
C) silver hydroxide
D) lead (II) hydroxide
E) iron (II) hydroxide
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell is made by placing an iron electrode in a compartment in which the Fe2+ concentration is 2.0 × 10-5 M and by placing a Pt electrode in the other compartment,in which the H+ concentration is 3.4 M and 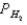 = 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is the value of E° for this cell,and which electrode is the anode?
= 1.00 atm.The Fe2+/Fe half-cell reduction potential is -0.41 V,and the H+/H2 half-cell reduction potential is 0.00 V.What is the value of E° for this cell,and which electrode is the anode?
A) 0.41 V,Fe
B) 0.90 V,Pt
C) -0.41 V,Pt
D) -0.41 V,Fe
E) 0.41 V,Pt
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a galvanic cell using Fe | Fe2+(1.0 M) and Pb | Pb2+(1.0 M) half-cells,which of the following statements is correct? Fe2+(aq) + 2e- → Fe(s) ; E° = -0.41 V Pb2+(aq) + 2e- → Pb(s) ; E° = -0.13 V
A) The iron electrode is the cathode.
B) When the cell has completely discharged,the concentration of Pb2+ is zero.
C) The mass of the iron electrode increases during discharge.
D) The concentration of Pb2+ decreases during discharge.
E) Electrons leave the lead electrode to pass through the external circuit during discharge.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The _____ has zinc can as the anode; a graphite rod in the center,surrounded by a paste of manganese dioxide,ammonium and zinc chlorides,and carbon black,is the cathode.
A) Poggendorff acid cell
B) Bunsen cell
C) Leclanché dry cell
D) Daniell cell
E) Weston cell
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the cell reaction for the following electrochemical cell? Zn | Zn2+(aq) || Sc3+(aq) | Sc
A) 3Zn(s) + 2Sc3+(aq) → 2Sc(s) + 3Zn2+(aq)
B) 2Sc(s) + 3Zn2+(aq) → 3Zn(s) + 2Sc3+(aq)
C) Zn(s) + Zn2+(aq) → Sc(s) + Sc3+(aq)
D) Zn(s) + Sc3+(aq) → Sc(s) + Zn2+(aq)
E) Sc(s) + Zn2+(aq) → Zn(s) + Sc3+(aq)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given:
W3+(aq) + 3e-  W(s) ; E° = 2.72 V
Pb2+(aq) + 2e-
W(s) ; E° = 2.72 V
Pb2+(aq) + 2e-  Pb(s) ; E° = -0.13 V
Ni2+(aq) + 2e-
Pb(s) ; E° = -0.13 V
Ni2+(aq) + 2e-  Ni(s) ; E° = -0.23 V
Cd2+(aq) + 2e-
Ni(s) ; E° = -0.23 V
Cd2+(aq) + 2e-  Cd(s) ; E° = -0.40 V
Zn2+(aq) + 2e-
Cd(s) ; E° = -0.40 V
Zn2+(aq) + 2e-  Zn(s) ; E° = -0.76 V
Al3+(aq) + 3e-
Zn(s) ; E° = -0.76 V
Al3+(aq) + 3e-  Al(s) ; E° = -1.66 V
Mg2+(aq) + 2e-
Al(s) ; E° = -1.66 V
Mg2+(aq) + 2e-  Mg(s) ; E° = -2.38 V
Under standard-state conditions,which of the following metals will reduce W3+ to W but will not reduce Ni2+ to Ni?
Mg(s) ; E° = -2.38 V
Under standard-state conditions,which of the following metals will reduce W3+ to W but will not reduce Ni2+ to Ni?
A) Cd
B) Pb
C) Al
D) Zn
E) Mg
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The process of producing a chemical change in an electrolytic cell is called _____.
A) electrodialysis
B) electrophoresis
C) electroplating
D) electrodeposition
E) electrolysis
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given:
Mn2+(aq) + 2e-  Mn(s) ; E° = -1.18 V
Cu2+(aq) + 2e-
Mn(s) ; E° = -1.18 V
Cu2+(aq) + 2e-  Cu(s) ; E° = 0.34 V
Cr2O72-(aq) + 14H+(aq) + 6e-
Cu(s) ; E° = 0.34 V
Cr2O72-(aq) + 14H+(aq) + 6e-  2Cr3+(aq) + 7H2O(l) ; E° = 1.33 V
Which of the following species is the strongest reducing agent?
2Cr3+(aq) + 7H2O(l) ; E° = 1.33 V
Which of the following species is the strongest reducing agent?
A) Mn
B) Cu
C) Cr3+
D) Mn2+
E) Cr2O72-
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the cell is initially at standard-state conditions,which of the following statements is true? 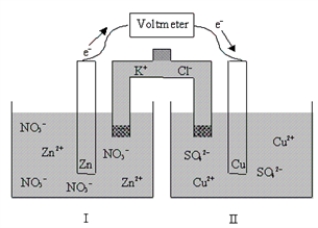 Zn2+(aq) + 2e-
Zn2+(aq) + 2e-  Zn(s) ; E° = -0.76 V
Cu2+(aq) + 2e-
Zn(s) ; E° = -0.76 V
Cu2+(aq) + 2e-  Cu(s) ; E° = 0.34 V
Cu(s) ; E° = 0.34 V
A) Initially Kc = 2 × 10-37,and it decreases with time.
B) Initially Kc = 2 × 1037,and it does not change with time.
C) Initially Kc = 2 × 1037,and it decreases with time.
D) Initially Kc = 2 × 1037,and it increases with time.
E) Initially Kc = 2 × 10-37,and it increases with time.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is one of the major products in the electrolysis of a saturated aqueous sodium chloride solution?
Reduction Half-Reaction
E° (V)
Na+(aq) + e-  Na(s) -2.71
Mg2+(aq) + 2e-
Na(s) -2.71
Mg2+(aq) + 2e-  Mg(s) -2.37
2H2O(l) + 2e-
Mg(s) -2.37
2H2O(l) + 2e-  H2(g) + 2OH-(aq) -0.83
O2(g) + 4H+(aq) + 4e-
H2(g) + 2OH-(aq) -0.83
O2(g) + 4H+(aq) + 4e-  2H2O(l) 1.23
Cl2(g) + 2e-
2H2O(l) 1.23
Cl2(g) + 2e-  2Cl-(aq)
1.36
2Cl-(aq)
1.36
A) Mg
B) O2
C) H2O
D) Na
E) Cl2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the cell reaction for the following voltaic cell? Cr(s) | Cr3+(aq) || Br-(aq) | Br2(g) | Pt(s)
A) 2Cr(s) + 3Br2(g) ![]() 2Cr3+(aq) + 6Br-(aq)
2Cr3+(aq) + 6Br-(aq)
B) Cr(s) + Cr3+(aq) ![]() Br-(aq) + Br2(g)
Br-(aq) + Br2(g)
C) 2Cr3+(aq) + 6Br-(aq) ![]() 2Cr(s) + 3Br2(g)
2Cr(s) + 3Br2(g)
D) Cr(s) + 3Br2(g) ![]() Cr3+(s) + 2Br-(aq)
Cr3+(s) + 2Br-(aq)
E) Cr(s) + 2Br-(aq) ![]() Br2(g) + Cr3+(aq)
Br2(g) + Cr3+(aq)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following electrochemical cell,what is the reduction half reaction? Mn(s) | Mn2+(aq) || Fe3+(aq) ,Fe2+(aq) | Pt(s)
A) Fe3+(aq) + e− → Fe2+(aq)
B) Fe2+(aq) + e− → Fe3+(aq)
C) Fe2+(aq) + Pt(s) → Fe3+(aq) + e−
D) Mn2+(aq) → Mn(s) + 2e−
E) Mn(s) → Mn2+(aq) + 2e−
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following oxidation-reduction reaction in acidic solution is balanced,what is the lowest whole-number coefficient for H+,and on which side of the balanced equation should it appear? S2O82-(aq) + NO(g) → SO42-(aq) + NO3-(aq)
A) 4,product side
B) 8,reactant side
C) 12,reactant side
D) 8,product side
E) 4,reactant side
G) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Showing 1 - 20 of 122
Related Exams