Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which contains 7 neutrons?
A) ![]() C
C
B) ![]() N
N
C) ![]() B
B
D) ![]() Si
Si
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which group do all atoms have seven valence electrons in the ground state?
A) alkali metals
B) alkaline earth metals
C) halogens
D) noble gases
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which orbital will have the lowest energy?
A) 4d
B) 4f
C) 4p
D) 4s
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope of hydrogen that has two neutrons is called
A) deuterium.
B) hydrogen.
C) dihydrogen.
D) tritium.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The nuclear model of the atom was constructed from which fundamental experiment?
A) alpha particle scattering by thin metal foils
B) cathode ray behavior in electrical fields
C) atomic spectra from gas discharge tubes
D) electrolysis of water
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Excited state electrons lose energy when they move to the ground state.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the particle that is the least penetrating.
A) alpha, α
B) beta, β
C) gamma, γ
D) delta, δ
F) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Alkali metals have an ns 2 outer electron structure.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many nucleons are there in the sulfur-35 isotope?
A) 16
B) 35
C) 19
D) 51
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cathode rays are beams of
A) anions.
B) electrons.
C) neutrons.
D) protons.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Isotopes are atoms of the same element with
A) different numbers of neutrons.
B) different numbers of electrons.
C) different numbers of protons.
D) different atomic numbers.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Tritium is the common name for
A) hydrogen-0.
B) hydrogen-1.
C) hydrogen-2.
D) hydrogen-3.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the same number of valence electrons as phosphorus?
A) Rb
B) Bi
C) In
D) Te
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metalloid
A) is the same as a metal.
B) is located on the left side of the periodic table.
C) has properties intermediate between metals and nonmetals.
D) is in the center of the periodic table.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Human beings use as much energy in one year as the amount of solar energy that hits the surface of the earth in
A) one hour.
B) one day.
C) one week.
D) one month.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The nuclear nature of the atom was postulated by
A) Rutherford.
B) Dalton.
C) Thomson.
D) Millikan.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom represented by 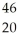 X has
X has
A) 20 protons and 26 neutrons.
B) 20 neutrons and 26 protons.
C) 20 protons and 46 neutrons.
D) 20 neutrons and 46 protons.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which radioactive particle was used by Geiger and Marsden in their experiments with thin metal foils?
A) alpha, α
B) beta, β
C) gamma, γ
D) lambda, λ
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which experimental observation led scientists to speculate that cathode rays contained particles that were constituents of all matter?
A) The rays were deflected by electrical fields.
B) The rays were deflected by magnetic fields.
C) The rays were independent of the type of gas in the vacuum tube.
D) All of the above provided evidence of the "fundamental" nature of cathode rays.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 190
Related Exams